Sulfuric Acid
Sulfuric acid, often referred to as the “universal chemical” or “the chemical that makes chemicals,” is a fundamental substance in chemical manufacturing. It serves as a key raw material or processing agent across numerous industries, including fertilizers, precious metal extraction, steel, paper, sugar, gasoline production, and water purification. This colorless, odorless liquid is highly reactive with water and organic materials, making it indispensable in chemical processes worldwide.
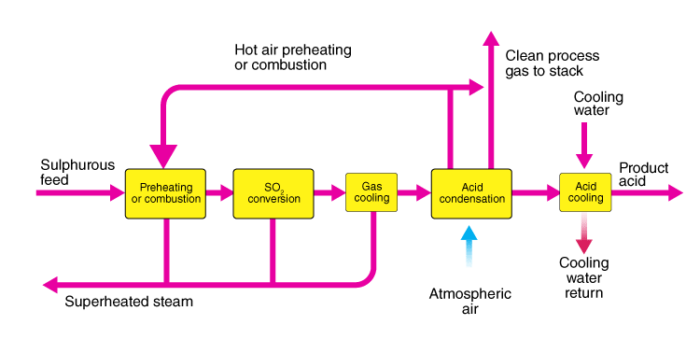
At Musa Pak Chemical Industries (MPCI), sulfuric acid is produced by the combustion of elemental sulfur (S) to form sulfur dioxide (SO₂), which is then oxidized catalytically to sulfur trioxide (SO₃). The SO₃ is subsequently absorbed in concentrated sulfuric acid (H₂SO₄) to form the final product.
MPCI possesses extensive experience in the safe handling and transportation of sulfuric acid. The acid is supplied in HM HDPE Carboys (50 kg) or in bulk via 10–15 metric ton road tankers, ensuring secure and timely delivery to clients.
Key Advantages
- Cost-effective production and procurement
- Low volatility
- Strong oxidizing agent
- Powerful dehydrating agent
- Highly acidic properties
Product Specifications
Physical Properties
- Appearance: Colorless liquid
- Specific Gravity (30°C): 1.82
- Boiling Point: 340°C
Chemical Specifications
- Purity (H₂SO₄): ≥ 98.0%
- Iron (Fe): ≤ 0.05%
- Residue on Ignition: ≤ 0.20%
- Lead (Pb): ≤ 0.005%
- Arsenic (As): ≤ 0.004%
Availability
Sulfuric acid is available in HM HDPE Carboys (50 kg) or in bulk via 10–15 metric ton road tankers. Delivery is customized to client requirements.
Handling & Storage
- Avoid contact with skin, eyes, and clothing.
- Do not inhale vapors or mist; use respiratory protection in poorly ventilated areas.
- Wash thoroughly after handling. Never add water directly to acid.
- Store in vented containers, away from combustible materials, metals, water, and alkalis.
- Containers must be specially designed for sulfuric acid.
First Aid Measures
Skin Contact: Flush with water for 15 minutes and remove contaminated clothing. Seek immediate medical attention.
Eye Contact: Rinse with water for at least 15 minutes. Keep eyelids away from eyeball. Seek immediate medical attention.
Inhalation: Move to fresh air. Provide rescue breathing if necessary. Keep warm and at rest. Seek medical attention.
Ingestion: If conscious, dilute with 1–3 glasses of water. Do not induce vomiting unless directed by medical personnel. Seek immediate medical attention.